Novartis AG – Annual report – 31 December 2019
Industry: pharmaceuticals
Novartis Group
consolidated financial statements (extract)
1. Significant accounting policies (extract)
Distribution of Alcon Inc. to Novartis AG shareholders
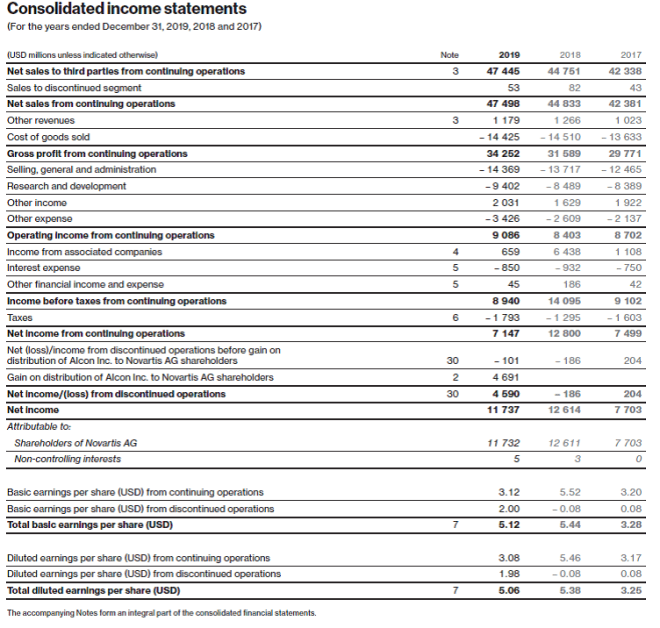
During the first quarter of 2019, at the Annual General Meeting (AGM) of Novartis AG shareholders, held on February 28, 2019, the Novartis AG shareholders approved a special distribution by way of a dividend in kind to effect the spin-off of Alcon Inc.
The February 28, 2019, shareholder approval for the spin-off required the Alcon Division and selected portions of corporate activities attributable to Alcon’s business (the “Alcon business”) to be reported as discontinued operations.
The shareholder approval to spin off the Alcon business also required the recognition of a distribution liability at the fair value of the Alcon business. The Group elected to measure the distribution liability at the fair value of the Alcon business net assets taken as a whole. The distribution liability was recognized through a reduction in retained earnings. It was required to be adjusted at each balance sheet date for changes in its estimated fair value, up to the date of the distribution to shareholders through retained earnings. Any resulting impairment of the business assets to be distributed would have been recognized in the consolidated income statements in “Other expense” of discontinued operations, at the date of initial recognition of the distribution liability or at subsequent dates resulting from changes of the distribution liability valuation. At the April 8, 2019 distribution settlement date, the resulting gain, which was measured as the excess amount of the distribution liability over the then-carrying value of the net assets of the business distributed, was recognized on the line “Gain on distribution of Alcon Inc. to Novartis AG shareholders” in the income statement of discontinued operations.
The recognition of the distribution liability required the use of valuation techniques for purposes of impairment testing of the Alcon business’ assets to be distributed and for the measurement of the fair value of the distribution liability. These valuations required the use of management assumptions and estimates related to the Alcon business’ future cash flows, market multiples to estimate day one market value, and control premiums to apply in estimating the Alcon business fair value. These fair value measurements were classified as “Level 3” in the fair value hierarchy. The section “—Impairment of goodwill and intangible assets” in this Note 1 provides additional information on key assumptions that are highly sensitive in the estimation of fair values using valuation techniques.
Transaction costs that were directly attributable to the distribution (spin-off) of Alcon to the Novartis shareholders, and that would otherwise have been avoided, were recorded as a deduction from equity.
For additional disclosures, refer to “Note 2. Significant transactions—Significant transactions in 2019— Completion of the spin-off of the Alcon business through a dividend in kind distribution to Novartis AG shareholders,” and “Note 30. Discontinued operations.”
2. Significant transactions (extract)
Significant transactions in 2019 (extract)
Completion of the spin-off of the Alcon business through a dividend in kind distribution to Novartis AG shareholders
On June 29, 2018, Novartis announced its intention to seek shareholder approval for the spin-off of the Alcon business into a separately traded standalone company, following the complete structural separation of the Alcon business into a standalone company (the Alcon business or Alcon Inc.).
The Novartis AG shareholders approved the spin-off of the Alcon business at the 2019 Annual General Meeting held on February 28, 2019, subject to completion of certain conditions precedent to the distribution. Upon shareholder approval, the Alcon business was reported as discontinued operations, and the fair value of the Alcon business exceeded the carrying value of its net assets.
The conditions precedent to the spin-off were met and on April 8, 2019 the spin-off of the Alcon business was effected by way of a distribution of a dividend in kind of Alcon Inc. shares to Novartis AG shareholders and ADR (American Depositary Receipt) holders (the Distribution), which amounted to USD 23.4 billion and is recognized as a reduction to retained earnings. Through the Distribution, each Novartis AG shareholder received one Alcon Inc. share for every five Novartis AG shares/ADRs they held on April 8, 2019, close of business. As of April 9, 2019, the shares of Alcon Inc. are listed on the SIX Swiss Exchange (SIX) and on the New York Stock Exchange (NYSE) under the symbol “ALC.”
The dividend in kind distribution liability to effect the spin-off of the Alcon business (the distribution liability) amounted to USD 26.4 billion at March 31, 2019, unchanged from its initial recognition on February 28, 2019, and was in excess of the carrying value of the Alcon business net assets as of February 28, 2019, and as of March 31, 2019. The net assets of the Alcon business amounted to USD 23.1 billion as at March 31, 2019.
On March 6, 2019, Alcon entered into financing arrangements with a syndicate of banks under which it borrowed on April 2, 2019, a total amount of USD 3.2 billion. These borrowings consisted of approximately USD 2.8 billion and the equivalent of USD 0.4 billion in EUR in bridge and other term loans under such Alcon facilities agreement. In addition, approximately USD 0.3 billion of borrowings under a number of local bilateral facilities in different countries, with the largest share of borrowings in Japan, were raised. This resulted in a total gross debt of USD 3.5 billion. These outstanding borrowings of the Alcon legal entities were recorded in the balance sheet and financing cash flow from discontinued operations. Prior to the spin-off, through a series of intercompany transactions, Alcon legal entities paid approximately USD 3.1 billion in cash to Novartis and its affiliates.
At the April 8, 2019 Distribution, the fair value of the distribution liability of the Alcon business amounted to USD 23.4 billion, a decrease of USD 3.0 billion from March 31, 2019. As mentioned above, prior to the spinoff, through a series of intercompany transactions, Alcon legal entities incurred additional net financial debt and paid approximately USD 3.1 billion in cash to Novartis and its affiliates. This additional net debt and transactions resulted in a decrease in Alcon’s net assets to USD 20.0 billion at the date of the Distribution of the dividend in kind to Novartis AG shareholders on April 8, 2019. The distribution liability at April 8, 2019, remained in excess of the then-carrying value of the Alcon business net assets.
Certain consolidated foundations own Novartis AG dividend-bearing shares restricting their availability for use by the Group. These Novartis AG shares are accounted for as treasury shares. Through the Distribution, these foundations received Alcon Inc. shares representing an approximate 4.7% equity interest in Alcon Inc. Upon the loss of control of Alcon Inc. through the Distribution, the financial investment in Alcon Inc. was recognized at its fair value based on the opening traded share price of Alcon Inc. on April 9, 2019 (a Level 1 hierarchy valuation). At initial recognition, its fair value of USD 1.3 billion was reported on the Group’s consolidated balance sheet as a financial asset. Management has designated this investment at fair value through other comprehensive income.
The total non-taxable, non-cash gain recognized at the distribution date of the spin-off of the Alcon business amounted to USD 4.7 billion consisting of:

1 See Note 30 for additional information.
For additional disclosures on discontinued operations, refer to Note 30.
30. Discontinued operations
Discontinued operations include the operational results from the Alcon eye care devices business and certain Corporate activities attributable to the Alcon business prior to the spin-off, the gain on distribution of Alcon Inc. to Novartis AG shareholders, and certain other expenses related to the Distribution (refer to Notes 1 and 2 for further details).
The Alcon eye care devices business researched, discovered, developed, manufactured, distributed and sold a broad range of eye care products. Alcon was organized into two global business franchises, Surgical and Vision Care. Alcon also provided services, training, education and technical support for both the Surgical and Vision Care businesses.

1 The consolidated income statement amounts are for the period from January 1, 2019, to the completion of the spin-off.
2 See Note 2 for further details on the non-taxable non-cash gain on distribution of Alcon Inc. to Novartis AG shareholders.
Supplemental disclosures related to the Alcon business distributed to Novartis AG shareholders
Net income
Included in net income from discontinued operations are:

1 Depreciation of right-of-use assets recognized from January 1, 2019, the date of implementation of IFRS 16 leases. See Note 1 for additional disclosures.
2 2018 includes an impairment of USD 337 million related to the write-down of the CyPass currently marketed product, which was acquired with the Alcon Division 2016 acquisition of Transcend Medical, Inc.
Balance sheet
The following were in the balance sheet from discontinued operations for the period from January 1, 2019, to the date of reclassification:

1 Additions to right-of-use assets recognized in 2019 with the implementation of IFRS 16 Leases on January 1, 2019. See Note 1 for additional disclosures.
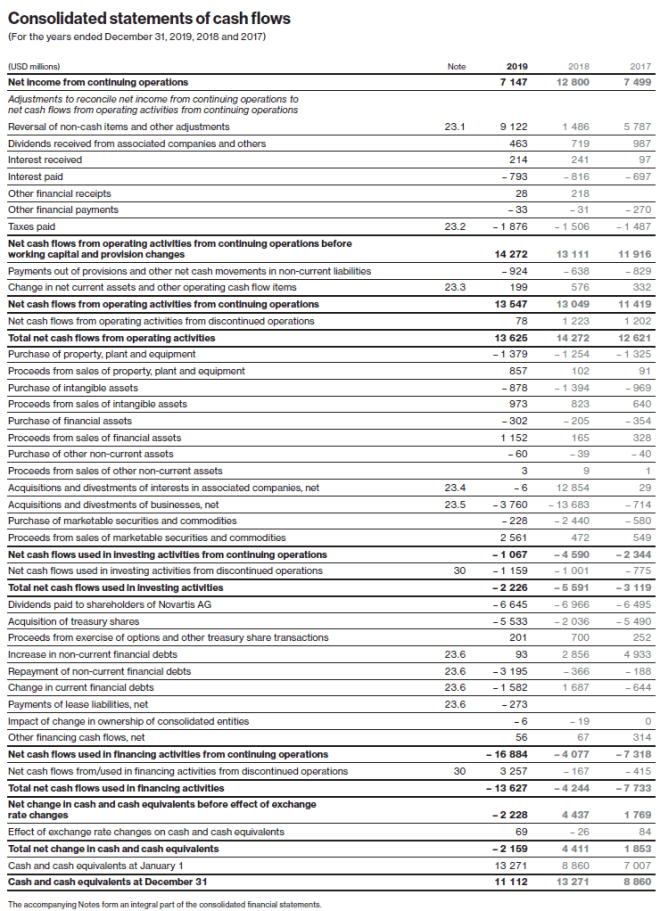
Cash flows used in investing activities from discontinued operations
Cash flows used in investing activities from discontinued operations include the investing activities of the Alcon business, and in addition in 2017 USD 140 million cash outflows for transaction-related expenditures attributable to the series of portfolio transformation transactions completed in 2015.

Cash flows from financing activities from discontinued operations
In 2019, the net cash inflows from financing activities from discontinued operations of USD 3.3 billion (2018: USD 167 million net cash outflows, 2017: USD 415 million net cash outflows) included USD 3.5 billion cash inflows from borrowings in connection with the distribution (spinoff) of the Alcon business to Novartis AG shareholders and USD 212 million (2018: USD 57 million, 2017: nil) transaction cost payment directly attributable to the distribution (spin-off) of the Alcon business to Novartis shareholders (see Notes 1 and 2).
Intangible assets
The Alcon Divisions’ cash-generating units, to which goodwill is allocated, each comprise a group of smaller cash-generating units.
The valuation method of the recoverable amount of the cash-generating units, to which goodwill is allocated, is based on the fair value less costs of disposal.
In 2017, the Alcon brand name indefinite life intangible asset was reported in Corporate, as it was used to market products of the Alcon Division and products within the Ophthalmology business franchise of the Innovative Medicines Division. In connection with the spin-off of the Alcon Division, the Novartis Group transferred the full rights of the Alcon brand name to the Alcon Division. As a result, the Innovative Medicines Division started the process of rebranding the products within its Ophthalmology business franchise and is no longer using the Alcon brand name. The Alcon brand name indefinite life intangible asset is therefore reported in the Alcon Division in 2018. In 2018, net sales of the Alcon Division products together are the grouping of cash-generating units, which were used to determine the recoverable amount. In the year before, net sales of products within the Innovative Medicines Ophthalmology business franchise as well as Alcon Division products, which used the Alcon brand name, together were the grouping of cash-generating units, which were used to determine the recoverable amounts. The valuation method is based on the fair value less costs of disposal.
The assumptions used in the calculations of fair value were a discount rate (post-tax) of 7.5% and a terminal growth rate of 3%. The Alcon terminal growth rate assumption of 3% is higher than the expected inflation rate of the medical device industry, and more specifically the ophthalmic sub-segment of the industry. The growth rates are expected to exceed this long-term inflation rate, as the aging population to which Alcon’s products are prescribed is growing faster than the general population. The discount rates consider the Group’s weighted average cost of capital, adjusted to approximate the weighted average cost of capital of a comparable market participant.
The fair value less costs of disposal, for all groupings of cash-generating units containing goodwill or indefinite life intangible assets, is reviewed for the impact of reasonably possible changes in key assumptions. In particular, we considered an increase in the discount rate, a decrease in the terminal growth rate, and certain negative impacts on the forecasted cash flows. These reasonably possible changes in key assumptions did not indicate an impairment.
“Note 1. Significant accounting policies—Impairment of goodwill and intangible assets” provides additional disclosures on how the Group performs goodwill and intangible asset impairment testing.
The 2018 intangible asset and goodwill impairment charges were USD 391 million, including an impairment of USD 337 million related to the write-down of the CyPass currently marketed product, which was acquired with the Alcon Division 2016 acquisition of Transcend Medical, Inc.
Leases
The following table shows the receivables of the gross investments in finance leases and the net present value of the minimum lease payments, as well as unearned finance income, related to Alcon’s surgical equipment lease arrangements. The finance income was recorded in “Other income.”

1 The current portion of the minimum lease payments was recorded in trade receivables or other current assets (to the extent not invoiced).
The lease liabilities recorded in discontinued operations on January 1, 2019, the date of implementation of IFRS 16 leases (see Note 1), were USD 286 million, and the right-of-use assets were USD 276 million, including USD 89 million and USD 75 million, respectively, for the previously reported finance lease obligations. For discontinued operations, there were no impairments or significant contract terminations of right-of-use assets for the period from January 1, 2019, to February 28, 2019, the date of shareholder approval for the Alcon spin-off.
Net assets derecognized
The following table presents the Alcon business net assets at the date of spin-off at April 8, 2019:

Defined contribution plans
In many subsidiaries, associates are covered by defined contribution plans. Contributions charged to the consolidated income statement for the defined contribution plans were:
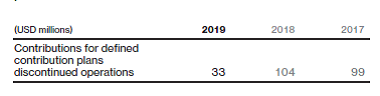
Significant transactions
In March 2019, Alcon acquired PowerVision, Inc. (PowerVision), a privately held, US-based medical device development company focused on developing accommodative, implantable intraocular lenses. The fair value of the total purchase consideration was USD 424 million. The amount consisted of an initial cash payment of USD 289 million and the net present value of the contingent consideration of USD 135 million, due to PowerVision shareholders, which they are eligible to receive upon the achievement of specified regulatory and commercialization milestones. The purchase price allocation resulted in net identifiable assets of USD 418 million, consisting of intangible assets of USD 505 million, net deferred tax liabilities of USD 93 million, other net assets of USD 6 million, and goodwill of USD 6 million. The 2019 results of operations since the date of the acquisition are not material.
For additional information related to the distribution (spin-off) of the Alcon business to Novartis AG shareholders, effected through a dividend in kind distribution that was completed on April 8, 2019, refer to Note 1 and Note 2.